Corranulene 1 is a bowl-shaped aromatic compound. It inverts through a planar transition state with a barrier of at 11.5 kcal mol-1. What changes would be found if one per-phenylated corranulene, making 2?
|
|
|
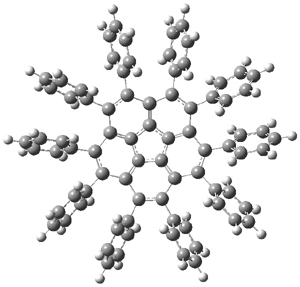
Scott1 has prepared 2a-c by arylating corranulene using phenylboroxin and palladium acetate and repeating this arylation four times. Amazing to me is that the yield of 2c is 54%! The BMK/cc-pVDZ optimized structure of 2a is shown in Figure 1. One can readily see that the bowl is nearly flat (click on the image to activate Jmol; the x-ray structure of 2b has the bowl depth of only 0.248 Å, compared to a depth of 0.87 Å in 1.
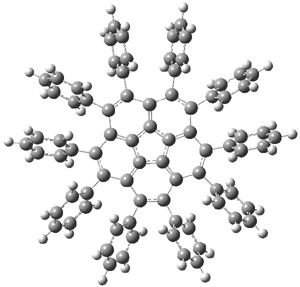
Interestingly, 2 inverts through a chiral TS (shown in Figure 1) so that inversion does not create the enantiomer! The computed barrier height is only 2.5 kcal mol-1.
|
2a |
2aTS |
Figure 1. BMK/cc-pVDZ optimized structures of 2a and the bowl inversion transition state 2aTS.
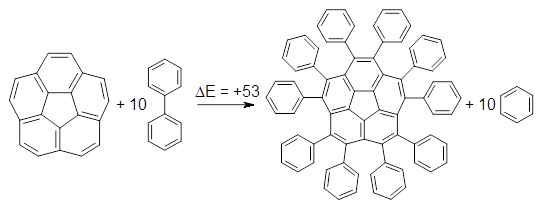
The flatter bowl results in longer bonds and wider angles about the rim of 2 than in 1. As one might expect, 2a is very strained: the BMK/cc-pVDZ estimation is that 2 is 53 kcal mol-1 more strained than 1, using the homodesmotic Reaction 1. In total, this is a real nice study of using strain to alter shape.
|
|
Reaction 1 |
References
(1) Zhang, Q.; Kawasumi, K.; Segawa, Y.; Itami, K.; Scott, L. T. "Palladium-Catalyzed C–H Activation Taken to the Limit. Flattening an Aromatic Bowl by Total Arylation," J. Am. Chem. Soc., 2012, 134, 15664-15667, DOI: 10.1021/ja306992k
InChIs
1: InChI=1S/C20H10/c1-2-12-5-6-14-9-10-15-8-7-13-4-3-11(1)16-17(12)19(14)20(15)18(13)16/h1-10H
InChIKey=VXRUJZQPKRBJKH-UHFFFAOYSA-N
2a: InChI=1S/C80H50/c1-11-31-51(32-12-1)61-62(52-33-13-2-14-34-52)72-65(55-39-19-5-20-40-55)66(56-41-21-6-22-42-56)74-69(59-47-27-9-28-48-59)70(60-49-29-10-30-50-60)75-68(58-45-25-8-26-46-58)67(57-43-23-7-24-44-57)73-64(54-37-17-4-18-38-54)63(53-35-15-3-16-36-53)71(61)76-77(72)79(74)80(75)78(73)76/h1-50H
InChIKey=OGBCICIRFDLAPN-UHFFFAOYSA-N
2b: InChI=1S/C120H130/c1-111(2,3)81-51-31-71(32-52-81)91-92(72-33-53-82(54-34-72)112(4,5)6)102-95(75-39-59-85(60-40-75)115(13,14)15)96(76-41-61-86(62-42-76)116(16,17)18)104-99(79-47-67-89(68-48-79)119(25,26)27)100(80-49-69-90(70-50-80)120(28,29)30)105-98(78-45-65-88(66-46-78)118(22,23)24)97(77-43-63-87(64-44-77)117(19,20)21)103-94(74-37-57-84(58-38-74)114(10,11)12)93(73-35-55-83(56-36-73)113(7,8)9)101(91)106-107(102)109(104)110(105)108(103)106/h31-70H,1-30H3
InChIKey=UQHBWLRORHEQNL-UHFFFAOYSA-N
2c: InChI=1S/C80H40Cl10/c81-51-21-1-41(2-22-51)61-62(42-3-23-52(82)24-4-42)72-65(45-9-29-55(85)30-10-45)66(46-11-31-56(86)32-12-46)74-69(49-17-37-59(89)38-18-49)70(50-19-39-60(90)40-20-50)75-68(48-15-35-58(88)36-16-48)67(47-13-33-57(87)34-14-47)73-64(44-7-27-54(84)28-8-44)63(43-5-25-53(83)26-6-43)71(61)76-77(72)79(74)80(75)78(73)76/h1-40H
InChIKey=MMASAVXWJRDYLD-UHFFFAOYSA-N

Henry Rzepa responded on 25 Oct 2012 at 12:01 pm #
The BMK functional is optimised for transition states, but it does not include dispersion terms. I cannot help wondering whether this system, with a large surface area of atoms pre-disposed for dispersion-style interactions, might be also studied with inclusion of this term, and whether that would influence the barrier for bowl-inversion?
Steven Wheeler responded on 04 Nov 2012 at 6:24 pm #
I’m not sure if inclusion of dispersion will strongly impact the bowl-inversion barrier, but I am certain it will reduce the computed strain energy significantly (I’m guessing by 10-20 kcal/mol).
Computational Organic Chemistry » Bowls derived from C70 responded on 06 Feb 2013 at 8:29 am #
[…] have discussed a few bowl-shaped aromatics in this blog (see for example this and this). Kuo and Wu now report on a few bowls derived from C70-fullerenes.1 The bowl 1 was […]