For the past twelve years, I have avoided posting on any of my own papers, but I will stoop to some shameless promotion to mention my latest paper,1 since it touches on some themes I have discussed in the past.
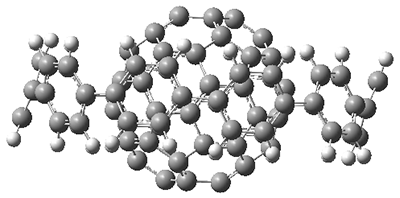
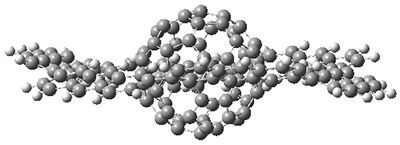
Back in 2011, Iwamoto, et al. prepared the complex of C60 1 surrounded by [10]cycloparaphenylene 2 to make the Saturn-like system 3.2 Just last year, Yamamoto, et al prepared the Nano-Saturn 5a as the complex of 1 with the macrocycle 4a.3 The principle idea driving their synthesis was to utilize a ring that is flatter than 2. The structures of 3 and 5b (made with the parent macrocycle 4b) are shown in side view in Figure 1, and clearly seen is the achievement of the flatter ring.

 3 |
5b |
7 |
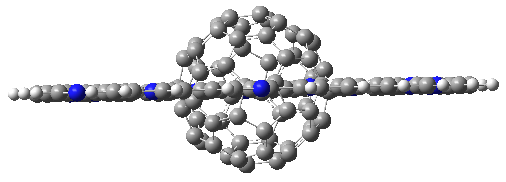
Figure 1. Computed structures of 3, 5, and 7.
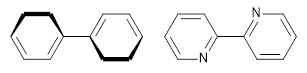
However, the encompassing ring is not flat, with dihedral angles between the anthrenyl groups of 35°. This twisting is due to the steric interactions of the ortho-ortho’ hydrogens. A few years ago, my undergraduate student David Stück and I suggested that selective substitution of a nitrogen for one of the C-H groups would remove the steric interaction,4 leading to a planar poly-aryl system, such as making twisted biphenyl into the planar 2-(2-pyridyl)-pyridine (Scheme 1)
Scheme 1.
Following this idea leads to four symmetrical nitrogen-substituted analogues of 4b; and I’ll mention just one of them here, 6.
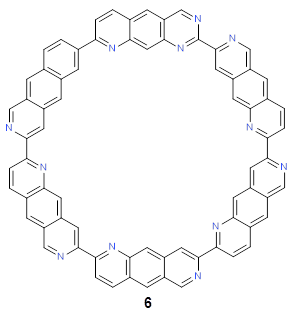
As expected, 6 is perfectly flat. The ring remains flat even when complexed with 1 (as per B3LYP-D3(BJ)/6-31G(d) computations), see the structure of 7 in Figure 1.
I also examined the complex of the flat macrocycle 6 (and its isomers) with a [5,5]-nanotube, 7. The tube bends over to create better dispersion interaction with the ring, which also become somewhat non-planar to accommodate the tube. Though not mentioned in the paper, I like to refer to 7 as Beyoncene, in tribute to All the Single Ladies.
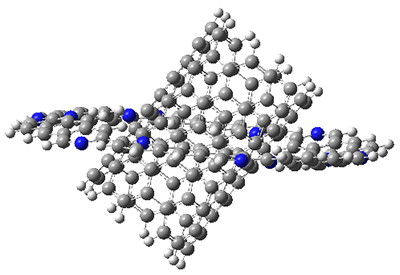
Figure 2. Computed structure of 7.
My sister is a graphic designer and she made this terrific image for this work:

References
1. Bachrach, S. M., “Planar rings in nano-Saturns and related complexes.” Chem. Commun. 2019, 55, 3650-3653, DOI: 10.1039/C9CC01234F.
2. Iwamoto, T.; Watanabe, Y.; Sadahiro, T.; Haino, T.; Yamago, S., “Size-Selective Encapsulation of C60 by [10]Cycloparaphenylene: Formation of the Shortest Fullerene-Peapod.” Angew. Chem. Int. Ed. 2011, 50, 8342-8344, DOI: 10.1002/anie.201102302
3. Yamamoto, Y.; Tsurumaki, E.; Wakamatsu, K.; Toyota, S., “Nano-Saturn: Experimental Evidence of Complex Formation of an Anthracene Cyclic Ring with C60.” Angew. Chem. Int. Ed. 2018 , 57, 8199-8202, DOI: 10.1002/anie.201804430.
4. Bachrach, S. M.; Stück, D., “DFT Study of Cycloparaphenylenes and Heteroatom-Substituted Nanohoops.” J. Org. Chem. 2010, 75, 6595-6604, DOI: 10.1021/jo101371m
InChIs
4b: InChI=1S/C84H48/c1-13-61-25-62-15-3-51-33-75(62)43-73(61)31-49(1)50-2-14-63-26-64-16-4-52(34-76(64)44-74(63)32-50)54-6-18-66-28-68-20-8-56(38-80(68)46-78(66)36-54)58-10-22-70-30-72-24-12-60(42-84(72)48-82(70)40-58)59-11-23-71-29-69-21-9-57(39-81(69)47-83(71)41-59)55-7-19-67-27-65-17-5-53(51)35-77(65)45-79(67)37-55/h1-48H
InChIKey=ZYXXLAYETADMDM-UHFFFAOYSA-N
6: InChI=1S/C72H36N12/c1-2-38-14-44-20-45-25-67(73-31-50(45)13-37(1)44)57-9-4-39-15-51-32-74-68(26-46(51)21-61(39)80-57)58-10-5-40-16-52-33-75-69(27-47(52)22-62(40)81-58)59-11-6-41-17-53-34-76-70(28-48(53)23-63(41)82-59)60-12-7-42-18-54-35-77-71(29-49(54)24-64(42)83-60)72-78-36-55-19-43-3-8-56(38)79-65(43)30-66(55)84-72/h1-36H
InChIKey=NSSCKPFBHGOOIJ-UHFFFAOYSA-N
