It never hurts to promote one’s science through clever names – think cubane, buckminsterfullerene, bullvalene, etc. Host-guest chemistry is not immune to this cliché too, and this post discusses the latest synthesis and computations of a nano-Saturn; nano-Saturns are a spherical guest molecule captured inside a ring host molecule. I discussed an example of this a number of years ago – the nano-Saturn comprised of C60 fullerene surrounded by [10]cycloparaphenylene.
Yamamoto, Tsurumaki, Wakamatsu, and Toyota have prepared a nano-Saturn complex with the goal of making a flatter ring component.1 The inner planet is modeled again by C60 and the ring is the [24]circulene analogue 1. The x-ray crystal structure of this nano-Saturn complex is shown in Figure 1.
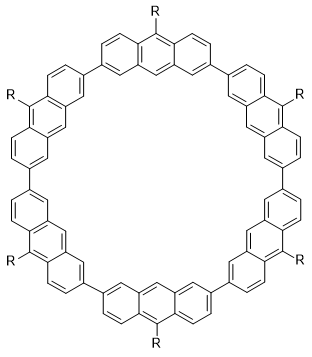
1: R = 2,4,6-tri-iso-propylphenyl
2: R = H
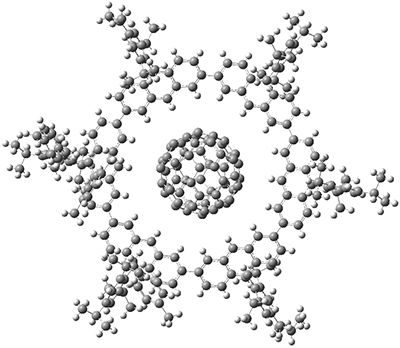
Figure 1. X-ray crystal structure of the nano-Saturn complex of 1 with C60.
Variable temperature NMR experiments gave the binding values of ΔH = -18.1 ± 2.3 kJ mol-1 and TΔS = 0.8 ± 2.2 kJ mol-1 at 298 K. To gauge this binding energy, they computed the complex of C60 with the parent compound 2 at B3LYP/6-1G(d)//M05-2X/6-31G(d), unfortunately without publishing the coordinates in the supporting materials. The computed binding enthalpy is ΔH = -50.6 kJ mol-1, but this computation is for the gas phase. The computed structure shows close contacts of 0.29 nm between the fullerene and the C9-proton of the anthracenyl groups, in excellent agreement with the x-ray structure. These weak C-H…π interactions undoubtedly help stabilize the complex, especially given that the fullerene carries a very tiny Mulliken charge of +0.08 e.
References
1) Yuta, Y.; Eiji, T.; Kan, W.; Shinji, T., "Nano-Saturn: Experimental Evidence of Complex Formation of an Anthracene Cyclic Ring with C60." Angew. Chem. Int. Ed. 2018, 57, 8199-8202, DOI: 10.1002/anie.201804430.
InChIs
1: InChI=1S/C174H180/c1-91(2)121-78-150(97(13)14)164(151(79-121)98(15)16)163-90-128-71-139-70-127-59-109(37-38-120(127)77-162(139)163)110-39-49-140-129(60-110)72-130-61-111(40-50-141(130)165(140)170-152(99(17)18)80-122(92(3)4)81-153(170)100(19)20)112-41-51-142-131(62-112)73-132-63-113(42-52-143(132)166(142)171-154(101(21)22)82-123(93(5)6)83-155(171)102(23)24)114-43-53-144-133(64-114)74-134-65-115(44-54-145(134)167(144)172-156(103(25)26)84-124(94(7)8)85-157(172)104(27)28)116-45-55-146-135(66-116)75-136-67-117(46-56-147(136)168(146)173-158(105(29)30)86-125(95(9)10)87-159(173)106(31)32)118-47-57-148-137(68-118)76-138-69-119(128)48-58-149(138)169(148)174-160(107(33)34)88-126(96(11)12)89-161(174)108(35)36/h37-108H,1-36H3
InChIKey=AMDNULXMAMDTMX-UHFFFAOYSA-N
2: InChI=1S/C84H48/c1-13-61-25-62-15-3-51-33-75(62)43-73(61)31-49(1)50-2-14-63-26-64-16-4-52(34-76(64)44-74(63)32-50)54-6-18-66-28-68-20-8-56(38-80(68)46-78(66)36-54)58-10-22-70-30-72-24-12-60(42-84(72)48-82(70)40-58)59-11-23-71-29-69-21-9-57(39-81(69)47-83(71)41-59)55-7-19-67-27-65-17-5-53(51)35-77(65)45-79(67)37-55/h1-48H
InChIKey=ZYXXLAYETADMDM-UHFFFAOYSA-N