Jacobsen has been pioneering the use of small organic molecules as catalysts where the interaction between the substrate and catalyst is non-covalent. At the 2009 Welch Conference last week, he spoke about the use of amido-thiourea catalysts in the enantioselective Strecker reaction. This post discusses how computations were used to determine the mechanism and helps explain the enantioselectivity.
The thiourea 1 was found to be the best catalyst for the hydrocyanation of imines. An example of this catalysis is Reaction 1. The system is quite tolerant to changes of the group attached to the carbon of the imine: with R = t-butyl group, the yield is 99% with an enantiomeric excess of 93% and with a phenyl group, the yield is 98% with 98% ee.1
|
|
Reaction 1 |
In a follow-up article, Jacobsen employs a nice combination of experiments and DFT computations to shed light on the mechanism.2 Kinetic studies established that the rate of the hydrocyanation is first order in HCN, imine and catalyst. This suggested two possible mechanisms, Scheme 1a (where protonation of the imine is followed by formation of the C-CN bond) and Scheme 1b (where cyanide adds first, followed by protonation).
Scheme 1.
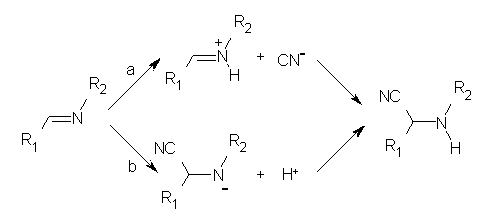
A Hammett study of phenyl-substituted imines indicates a negative ρ value, indicative of positive charge build up on nitrogen, suggesting the mechanism shown in Scheme 1a. Now on the computations!
A model study was first performed at B3LYP/6-31G(d) for the reaction of HCN with imine 2a with the catalyst 3a. Three complexes involving the three species of nearly equivalent energy were found. The first mechanism examined is for direct addition of cyanide to imine with the catalyst activating the imine. This pathway was identified but it has a very high barrier (46 kcal mol-1) and the rates of the urea and thiourea-catalyzed reactions are nearly identical, but the computations for this mechanism suggest that the thiourea-catalyzed reaction should be substantially faster. This mechanism can be discounted.

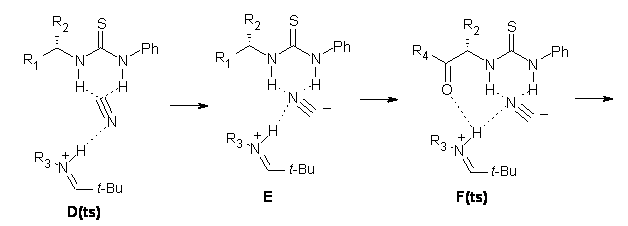
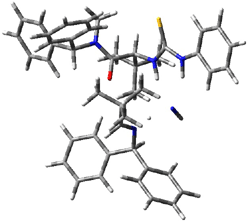
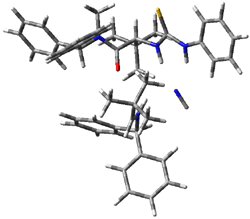
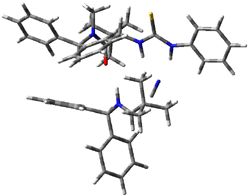
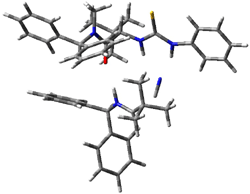
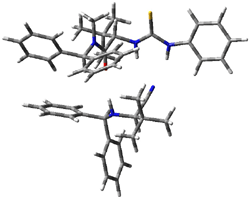
Next, they examined two related mechanism whereby either HCN or HNC complex to the catalysts and then transfer their proton to the imine. This is followed by cyanide attack on the protonated imine. The reaction involving HNC is slightly lower than that of HCN, and so this mechanism is pursued further with catalyst 3b, which more closely mimics the true catalyst, and imine 2b. The lowest energy pathway for the addition of HCN to 2b with catalyst 3b is schematically shown in Scheme 2. From A to E is transfer of the proton from HNC to the imine and rotation of the CN anion. The highest barrier is through TS F, which involves moving the imine over to the carbonyl group. The last step is the attack of the cyanide group. The structures of E through I are shown in Figure 1.
Scheme 2
|
|
|
|
|
|
|
E |
F |
|
G |
H |
|
I |
|
Figure 1. B3LYP/6-31G(d) optimized geometries for steps along Scheme 2.
The step involving the transition state F is rate determining. They then computed the difference in the activation barrier through TS F for a variety of different imines using the R– and S-catalyst and compared that with the experimental enantioselectivity. These turn out to correlate extremely well, suggesting that in fact the enantioselectivity is discriminated through TS F! And the structural feature that best correlates with this enantioselectivty is the sum of the O…H and N…H distances in F for the R– and S-catalysts. So it is the ability to stabilize the iminium cation that is key.
A couple of caveats: first, the potential energy surface of this reaction is quite complex – Jacobsen suggests a pathway with 9 critical points. One might imagine that there could be other pathways and other important critical points, and fully characterizing this surface would be an enormous task! The nice agreement between the computations and the available experiments do give the computed pathway some credence. Second, the reactions are run in solution, and solvent has been neglected in these computations. Given that charge-separated species are being formed, solvent could be a factor. Nonetheless, this remains an interesting set of papers as it shows how computations are now a standard tool even amongst synthetic chemists!
References
(1) Zuend, S. J.; Coughlin, M. P.; Lalonde, M. P.; Jacobsen, E. N., "Scaleable catalytic asymmetric Strecker syntheses of unnatural α-amino acids," Nature, 2009, 461, 968-970, DOI: 10.1038/nature08484.
(2) Zuend, S. J.; Jacobsen, E. N., "Mechanism of Amido-Thiourea Catalyzed Enantioselective Imine Hydrocyanation: Transition State Stabilization via Multiple Non-Covalent Interactions," J. Am. Chem. Soc. 2009, 131, 15358-15374, DOI: 10.1021/ja9058958.