What is the relationship between a ground state and the first excited triplet (or first excited singlet) state regarding aromaticity? Baird1 argued that there is a reversal, meaning that a ground state aromatic compound is antiaromatic in its lowest triplet state, and vice versa. It is suggested that the same reversal is also true for the second singlet (excited singlet) state.
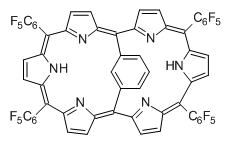
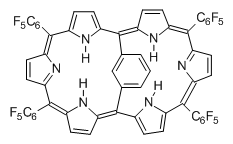
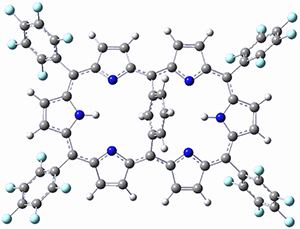
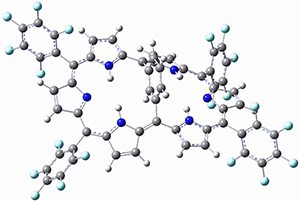
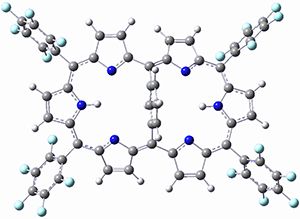
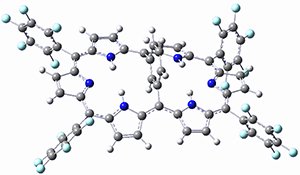
Osuka, Sim and coworkers have examined the geometrically constrained hexphyrins 1 and 2.2 1 has 26 electrons in the annulene system and thus should be aromatic in the ground state, while 2, with 28 electrons in its annulene system should be antiaromatic. The ground state and lowest triplet structures, optimized at B3LYP/6-31G(d,p), of each of them are shown in Figure 1.
|
|
|
|
11 |
12 |
|
31 |
32 |
Figure 1. B3LYP/6-31G(d,p) optimized geometries of 1 and 2.
NICS computations where made in the centers of each of the two rings formed by the large macrocycle and the bridging phenyl group (sort of in the centers of the two lenses of the eyeglass). The NICS values for 1 are about -15ppm, indicative of aromatic character, while they are about +15ppm for 2, indicative of antiaromatic character. However, for the triplet states, the NICS values change sign, showing the aromatic character reversal between the ground and excited triplet state. The aromatic states are also closer to planarity than the antiaromatic states (which can be seen by clicking on the images in Figure 1, which will launch the JMol applet so that you can rotate the molecular images).
They also performed some spectroscopic studies that support the notion of aromatic character reversal in the excited singlet state.
References
(1) Baird, N. C. "Quantum organic photochemistry. II. Resonance and aromaticity in the lowest 3ππ* state of cyclic hydrocarbons," J. Am. Chem. Soc. 1972, 94, 4941-4948, DOI: 10.1021/ja00769a025.
(2) Sung, Y. M.; Oh, J.; Kim, W.; Mori, H.; Osuka, A.; Kim, D. quot;Switching between Aromatic and Antiaromatic 1,3-Phenylene-Strapped [26]- and [28]Hexaphyrins upon Passage to the Singlet Excited State," J. Am. Chem. Soc. 2015, 137, 11856-11859, DOI: 10.1021/jacs.5b04047.
InChIs
1: InChI=1S/C60H18F20N6/c61-41-37(42(62)50(70)57(77)49(41)69)33-23-8-4-19(81-23)31-17-2-1-3-18(16-17)32(21-6-10-25(83-21)35(29-14-12-27(33)85-29)39-45(65)53(73)59(79)54(74)46(39)66)22-7-11-26(84-22)36(40-47(67)55(75)60(80)56(76)48(40)68)30-15-13-28(86-30)34(24-9-5-20(31)82-24)38-43(63)51(71)58(78)52(72)44(38)64/h1-16,85-86H/b31-19+,31-20+,32-21+,32-22+,33-23+,33-27+,34-24+,34-28+,35-25+,35-29+,36-26+,36-30+
InChIKey=TUOMWLSCXXODFY-CQGNQUHXSA-N
2: InChI=1S/C60H20F20N6/c61-41-37(42(62)50(70)57(77)49(41)69)33-23-8-4-19(81-23)31-17-2-1-3-18(16-17)32(21-6-10-25(83-21)35(29-14-12-27(33)85-29)39-45(65)53(73)59(79)54(74)46(39)66)22-7-11-26(84-22)36(40-47(67)55(75)60(80)56(76)48(40)68)30-15-13-28(86-30)34(24-9-5-20(31)82-24)38-43(63)51(71)58(78)52(72)44(38)64/h1-16,81-84H/b31-19+,31-20+,32-21+,32-22+,33-23+,33-27+,34-24+,34-28+,35-25+,35-29+,36-26+,36-30+
InChIKey=KTIAGNMFTAGKFJ-CQGNQUHXSA-N

Henry Rzepa responded on 26 Oct 2015 at 11:57 pm #
Re The aromatic states are also closer to planarity than the anti-aromatic states, it would be good to compute the relative contributions of twist and writhe to these intrinsically non-planar (lemniscular) systems as perhaps a better index, according to Lk = Tw + Wr. One might imagine that an aromatic system will twist less (and hence writhe more) compared to an anti-aromatic system, for which the reverse might be true (twisting being one way of relieving anti-aromaticity). Such an analysis on a different [26]-phyrin was in fact performed some years ago (doi: 10.1021/ol703129z), but it would be good to have further examples.
For twisted Möbius systems (systems with a linking number Lk ≠ 0) a generalisation of the rules for aromaticity has been noted (dos: 10.1016/j.comptc.2014.09.028).
Yunqi responded on 29 Oct 2015 at 9:52 pm #
Hi,
Sorry if it’s inappropriate to discuss this here, but I’ve encountered some problem trying to subscribe to this blog.
The images are all broken in the RSS feeds. I thought it might be due to some error when wordpress generates the image link ( a ‘blog/’ is missing in the url ).
I tried some other RSS readers and the problem strill exist, so I wonder if you could kindly fix this so that I can read this wonderful blog via RSS?
Thank you.
Yunqi responded on 30 Oct 2015 at 3:02 am #
Sorry for disturbing,
I’ve tried some more approaches and itturns out to be my problem.
Apology again!